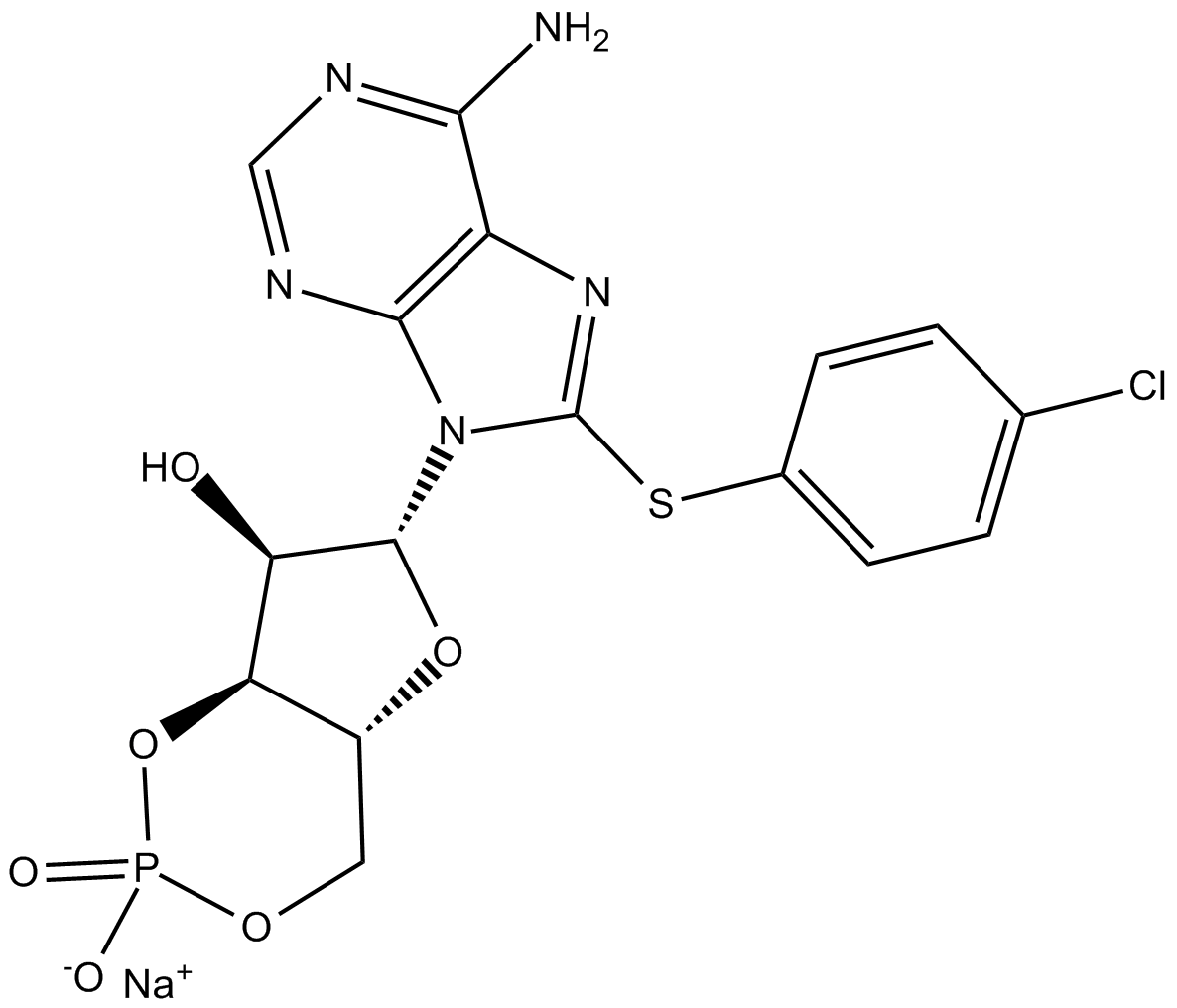
8-CPT-Cyclic AMP (sodium salt)
Ka : 0.05 and 0.11 μM for PKA and PKG, respectively
8-CPT-Cyclic AMP is a lipophilic activator of the cyclic-AMP- and cyclic-GMP-dependent protein kinases, PKA and PKG.
The ability of cGMP-dependent protein kinases (cGKs) to activate cAMP response element (CRE)-dependent gene transcription was compared with that of cAMP-dependent protein kinases (cAKs).
In vitro: A previous study found that 8-CPT-Cyclic AMP was a potent activator of cAMP-PK and cyclic-GMP-dependent protein kinase (cGMP-PK) both as purified enzymes and in platelet membranes. 8-CPT-Cyclic AMP was hydrolysed to a significant extent by the Ca2+/calmodulin-dependent phosphodiesterase and by the cGMP-inhibited phosphodiesterase. Moreover, Sp-5,6- DCl-cBiMPS was also more effective than 8-CPT-Cyclic AMP in inducing quantitative phosphorylation of vasodilator-stimulated phosphoprotein in intact platelets [1]. Another study indicated that 8-CPT-Cyclic AMP was a potent inhibitor of the cyclic GMP-specific phosphodiesterase (PDE VA). Indeed, 8-CPT-Cyclic AMP could inhibit PDE VA with a potency similar to that of zaprinast. In addition, 8-CPT-Cyclic AMP was metabolized by PDE VA at a rate half that of cyclic GMP. The cyclic GMP-inhibited phosphodiesterase and the cyclic AMP-specific phosphodiesterase could be inhibited by 8-CPT-Cyclic AMP as well [2].
In vivo: Up to now, there is no animal in vivo data reported.
Clinical trial: So far, no clinical study has been conducted.
References:
[1] Sandberg, M. ,Butt, E.,Nolte, C., et al. Characterization of Sp-5,6-dichloro-1-β-D-ribofuranosylbenzimidazole-3',5'-monophosphorothioate (Sp-5,6-DCl-cBiMPS) as a potent and specific activator of cyclic-AMP-dependent protein kinase in cell extracts and intact cells. Biochemistry Journal 279, 521-527 (1991).
[2] Connolly, B. J.,Willits, P.B.,Warrington, B.H., et al. 8-(4-chlorophenyl)thio-cyclic AMP is a potent inhibitor of the cyclic GMP-specific phosphodiesterase (PDE VA). Biochemical Pharmacology 44(12), 2303-2306 (1992).
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 493.8 |
| Cas No. | 93882-12-3 |
| Formula | C16H14ClN5O6PS·Na |
| Synonyms | 8-(p-Chlorophenylthio)-cAMP,8-CPT-cAMP |
| Solubility | ≤25mg/ml in H2O |
| Chemical Name | 8-[(4-chlorophenyl)thio]-cyclic 3',5'-(hydrogen phosphate)-adenosine, monosodium salt |
| SDF | Download SDF |
| Canonical SMILES | O[C@H]1[C@H](N2C(SC3=CC=C(Cl)C=C3)=NC4=C2N=CN=C4N)O[C@H]5[C@H]1OP(OC5)([O-])=O.[Na+] |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
-
Purity = 98.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
Chemical structure